ONTOZRY®▼ (cenobamate) Dosing & Titration

▼This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Adverse events and product complaint should be reported. Reporting forms and information can be found at www.mhra.gov.uk/yellowcard for the UK or www.hpra.ie for Ireland. Adverse events and product complaint should also be reported to Angelini Pharma on (UK) +44 2034889643, (IRE) +353 1 584 4671 or UKIReporting@angelinipharma.com
ⓘ Click here for Prescribing Information and Adverse Event Reporting
ONTOZRY® (cenobamate) tablets are taken once daily
ONTOZRY® (cenobamate) titration: dosing schedule
ONTOZRY® (cenobamate) missed doses and discontinuation
Special populations and ONTOZRY® (cenobamate) dosing
ONTOZRY® (cenobamate) interactions: dose adjustments
ONTOZRY® (cenobamate) tablets are taken once daily
ONTOZRY® tablets should be taken once daily, with or without food, and with a glass of water. The tablets can either be:1
- Swallowed whole
- Crushed and mixed with water
- Administered via a nasogastric (NG) tube
ONTOZRY® can be administered via nasogastric feeding as follows:1
- Crush the required number of tablets according to the prescribed dose.
- Place the crushed tablet(s) into a suitable container and add 25 mL of water.
- Shake the mixture to form a suspension.
- Using a syringe, transfer the suspension into the NG tube, ensuring the container is completely emptied.
- Refill the catheter-tip syringe with 10 mL of water, swirl gently, and flush through the NG tube.
- Check that no particles remain in the syringe. If any are visible, repeat the water flush in step 5.
ONTOZRY® should preferably be taken at the same time each day in the same way. The tablets should not be split, since there is no break line therefore the accuracy of the dose cannot be ensured.1
See the ONTOZRY® homepage for indication details.
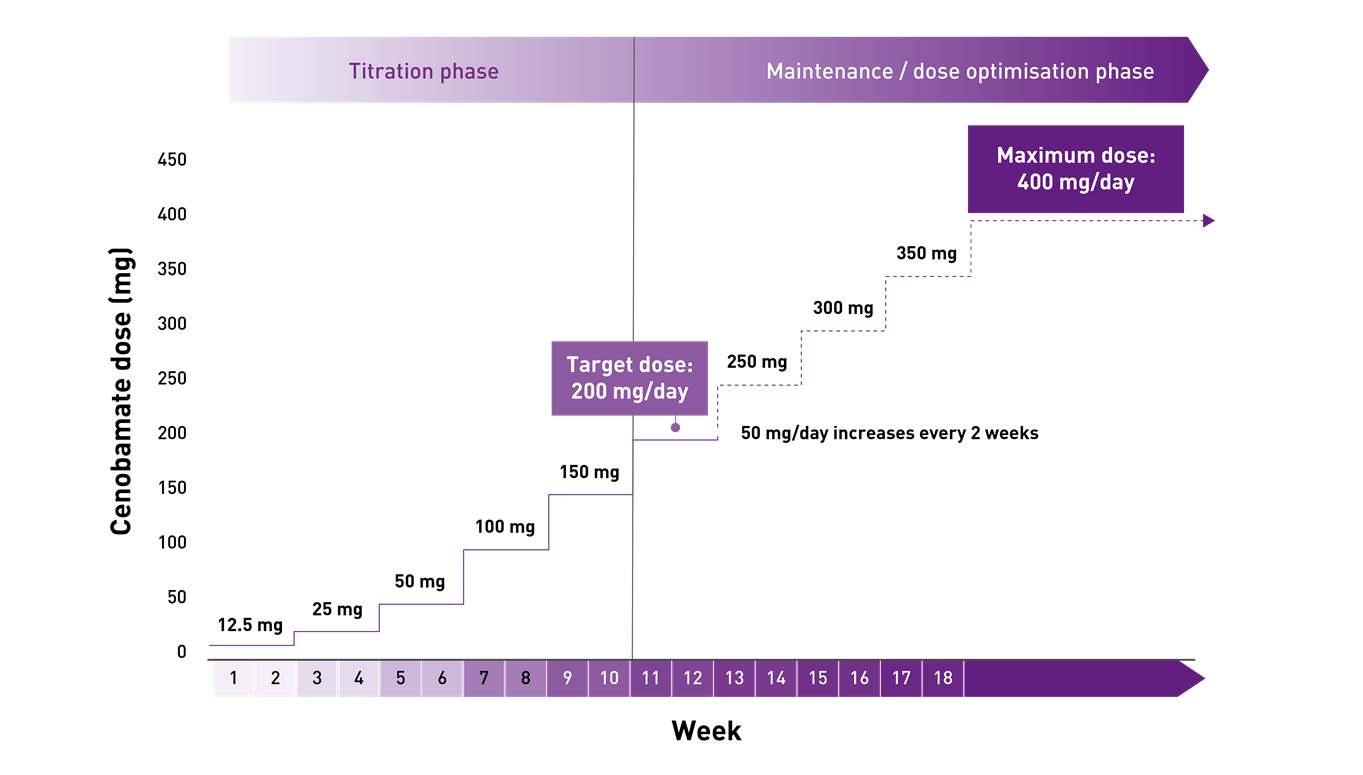
ONTOZRY® (cenobamate) titration: dosing schedule
The starting dose of ONTOZRY® is 12.5 mg once daily. It is then incrementally titrated up according to response, with each step of the titration phase recommended to last at least 2 weeks. The recommended target dose of ONTOZRY® is 200 mg/day. Based on clinical response, the dose may be increased to a maximum of 400 mg per day 1
For full guidance, see the ONTOZRY® (cenobamate) Clinical Guidelines.
ONTOZRY® (cenobamate) titration schedule for adults with focal-onset seizures in epilepsy1
| Treatment phase | Dose (per day, oral) | Weeks |
|---|---|---|
| Treatment initiation | 12.5 mg | 1 and 2 |
| 25 mg | 3 and 4 | |
| Titration | 50 mg | 5 and 6 |
| 100 mg | 7 and 8 | |
| 150 mg | 9 and 10 | |
| Target dose | 200 mg | 11, 12 and onwards |
| Dose optimisation | If optimal seizure control has not been achieved on the recommended target dose of 200 mg/day, patients may benefit from an increase in dose by 50 mg/day every 2 weeks up to the maximum dose of 400 mg/day. | |
The recommended titration schedule should not be exceeded because of the potential for serious adverse reactions
ONTOZRY® (cenobamate): initial dosing and initiation pack
ONTOZRY® comes in the form of an initiation pack that includes contains 14 tablets of 12.5 mg and 14 film-coated tablets of 25 mg doses to cover the treatment initiation over these first 4 weeks.1,2
Different dosages can be prescribed at the appropriate stages of the titration schedule, including 50 mg, 100 mg, 150 mg, and 200 mg.1
ONTOZRY® (cenobomate) slow titration: start low, go slow
The titration schedule has been established based on findings from relevant clinical trials and must not be exceeded due to the potential for serious adverse reactions.1
Findings from the Phase III, open-label, long-term safety study C021 support a start-low, go-slow titration for ONTOZRY®, starting at 12.5 mg once daily and up-titrating every two weeks over 12 weeks to 200 mg/day. This regime was associated with fewer and less frequent treatment-emergent adverse events (TEAEs) and a lower rate of Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) compared to the C017 study. Notably, no DRESS cases were reported among 1,339 patients in C021.3
In the Phase IIb, multicentre, randomised, double-blind, placebo-controlled efficacy and safety C017 study, a faster 6-week titration was used. The original regimen started at 100 mg/day with weekly 100 mg increments to target dose. An amendment to improve tolerability reduced the starting dose to 50 mg/day, with 50 mg weekly increases to 200 mg/day, then 100 mg weekly increases to 400 mg/day.4
Visit ONTOZRY® tolerability and side effects to better understand the TEAEs that a slower titration schedule may help to avoid.
ONTOZRY® (cenobamate) maintenance phase and dose optimisation
The maximum dose of ONTOZRY® is 400 mg/day. In the instances where patients do not reach optimal seizure control on the recommended dose of 200 mg/day, consider up-titrating by 50 mg/day every two weeks up to the maximum dose.1
In the C021 safety study, concomitant ASMs could be removed, added, or adjusted, and ONTOZRY® doses adjusted during the titration phase as clinically needed. Downward ASM dose adjustments could be made for tolerability during the maintenance phase for all patients once the target dose of 200 mg/day was reached.1,3
ONTOZRY® (cenobamate) missed doses and discontinuation
If a patient misses a dose, they should take a single dose as soon as they remember, unless it is less than 12 hours until their next regularly scheduled dose.1
If treatment needs to be stopped, discontinuation of ONTOZRY® should occur gradually to minimise the potential of rebound seizures over at least two weeks, unless safety concerns require abrupt withdrawal.1
Explore ONTOZRY® tolerability and side effects to better recognise when safety concerns may justify abrupt discontinuation.
Special populations and ONTOZRY® (cenobamate) dosing
Paediatric population
ONTOZRY® is not indicated for children aged 0 months to 18 years as no data is available to establish the safety and efficacy of ONTOZRY® in this population.1
Lactose intolerant
Patients with rare hereditary problems such as galactose intolerance, total lactase deficiency, or glucose-galactose malabsorption should not take the tablet.1
Elderly
Clinical studies of cenobamate did not include sufficient numbers of subjects aged 65 and over, to determine whether they responded differently from younger patients. It has been reported that elderly patients (65 years of age and above) have a higher risk of adverse reactions to antiepileptic drugs such as fatigue, gait disturbance, fall, ataxia, balance disorder, dizziness and somnolence. Dose selection in this population should be cautious, typically starting at the lower end of the recommended range. This increased susceptibility may be related to age-associated decline in hepatic and renal function, the presence of comorbidities, and the greater likelihood of drug interactions in polymedicated patients.1
Renal impairment
In patients with renal impairment, a reduction in target dose may be considered in patients with mild to moderate (creatinine clearance 30 to <90 ml/min) or severe (creatinine clearance < 30 ml/min) renal impairment and ONTOZRY® should be prescribed with caution. The maximum recommended dose of ONTOZRY® is 300mg/day for patients with mild to moderate (creatine clearance 30 to <90 ml/min) or severe (creatine clearance <30 ml/min) renal impairment.
ONTOZRY® should not be used in patients with end-stage renal disease or patients undergoing haemodialysis.1
Hepatic impairment
Patients with chronic hepatic disease experience increased cenobamate exposure due to decreased hepatic drug clearance. The maximum recommended dose may need to be decreased by up to 50%, with 200 mg/day as the recommended maximum dose in patients with mild or moderate hepatic impairment, however, the starting dose may remain the same. ONTOZRY® should not be used in patients with severe hepatic impairment.1,5
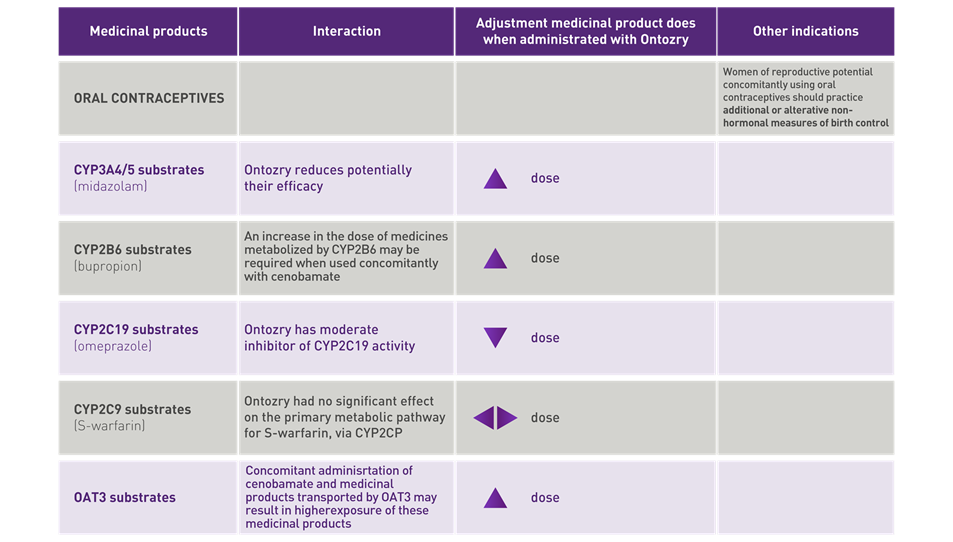
ONTOZRY® (cenobamate) interactions: dose adjustments
Cenobamate is extensively metabolized, primarily by glucuronidation, with oxidation contributing to a lesser degree. ONTOZRY® can interact with other medicinal products, due to its ability to induce or inhibit enzymes that belong to the Cytochrome P450 system, requiring adjustments of the dose of these medicinal products.
Some ASMs can also induce or inhibit Cytochrome P450 enzymes, affecting the plasma levels of concomitant drugs and their active metabolites. A slow titration schedule of ONTOZRY®, with proactive adjustment of concomitant ASMs, can help minimise TEAEs.5
No cenobamate dose adjustment needed with many concomitant ASMs1
Explore ONTOZRY® tolerability and side effects to understand the importance of preempting drug interactions and of aiming for proactive dose adjustment.
No dose adjustment is needed with many concomitant ASMs as part of the ONTOZRY® dosing schedule,1 though high retention rates were observed even after reducing the dose or completely discontinuing one or more concomitant ASMs.6
Cenobamate dose adjustments based on interactions with contraceptives and enzyme substrates1
Abbreviations
NG, nasogastric tube; TEAEs, treatment-emergent adverse events; DRESS, Drug Reaction with Eosinophilia and Systemic Symptoms; ASMs, anti-seizure medications; HCP, healthcare professional
- ONTOZRY® Summary of Product Characteristics. United Kingdom and European Union
- Ontozry Treatment Initiation Pack 12.5 mg tablets and 25 mg film-coated tablets - Summary of Product Characteristics (SmPC).
- Sperling MR, Klein P, Aboumatar S, et al. Cenobamate (YKP3089) as adjunctive treatment for uncontrolled focal seizures in a large, phase 3, multicenter, open-label safety study. Epilepsia. 2020;61(6):1099-1108.
- Steinhoff BJ, Ben-Menachem E, Brandt C, et al. Onset of efficacy and adverse events during Cenobamate titration period. Acta Neurol Scand. 2022;146(3):265-275.
- Schmitz B, Lattanzi S, Vonck K, Kälviäinen R, Nashef L, Ben-Menachem E. Cenobamate in refractory epilepsy: Overview of treatment options and practical considerations. Epilepsia Open. 2023;8(4):1241-1255.
- Rosenfeld WE, Abou-Khalil B, Aboumatar S, et al. Post hoc analysis of a phase 3, multicenter, open-label study of cenobamate for treatment of uncontrolled focal seizures: Effects of dose adjustments of concomitant antiseizure medications. Epilepsia. 2021;62(12):3016-3028.
MAT-UKI-0304-P | April 2025
 HarmoniaMentis
HarmoniaMentis
